Retin a 0.19/27/2023 INDICATION: Tretinoin Cream is indicated for topical application in the treatment of acne vulgaris. To report SUSPECTED ADVERSE REACTIONS contact FDA at fda.gov/medwatch or call 1-80. If these effects occur, treatment should be discontinued or adjusted to a level the patient can tolerate. Pregnancy: Tretinoin should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.Īdverse Reactions: The skin of certain sensitive individuals may become excessively red, edematous, blistered, or crusted. Tretinoin has been reported to cause severe irritation on eczematous skin and should be used with utmost caution in patients with this condition. 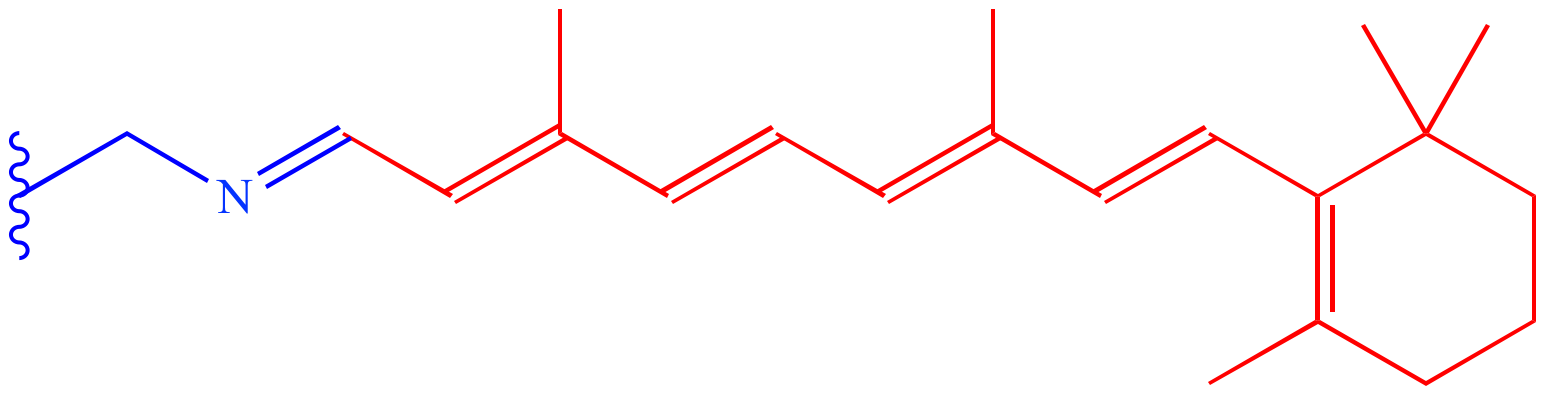
If the degree of local irritation warrants, patients should be directed to use the medication less frequently, discontinue use temporarily, or discontinue use altogether. Topical use may induce severe local erythema and peeling at the site of application. Tretinoin Gel should be kept away from the eyes, the mouth, angles of the nose, and mucous membranes. Weather extremes, such as wind or cold, also may be irritating to patients using tretinoin. Use of sunscreen products and protective clothing over treated areas is recommended when exposure cannot be avoided. 
Patients who have considerable sun exposure due to occupation and those with inherent sensitivity to the sun should exercise particular caution. 
Precautions: Exposure to sunlight, including sunlamps, should be minimized during the use of Tretinoin Cream and Gel, and patients with sunburn should be advised not to use the product until fully recovered. Please see full Prescribing Information for Tretinoin Gel INDICATION: Tretinoin Gel is indicated for topical application in the treatment of acne vulgaris.ĬONTRAINDICATION: Use of the product should be discontinued if hypersensitivity to any of the ingredients is noted.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
