Free nerve endings signal9/26/2023 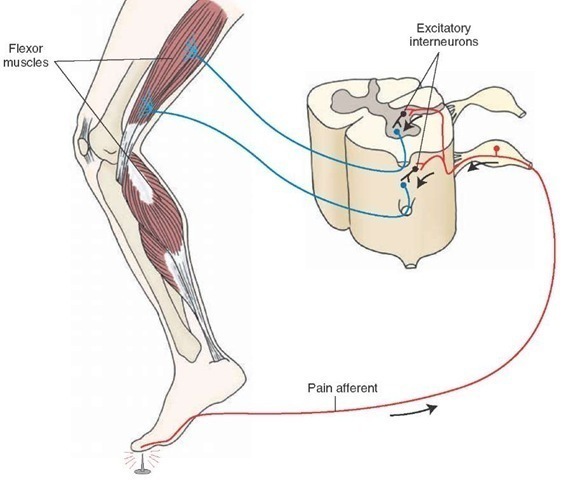
Na v1.7 loss-of-function mutations have been linked to inability to feel pain in humans. These channels shaping action potential generation and are critical for nociceptor neuron depolarization, thus mediating the initiation of pain signaling. Na v1.7, Na v1.8, and Na v1.9 are voltage-gated sodium channels enriched in nociceptor neurons compared to other neuronal subtypes. Nociceptive Ion Channels as Molecular Transducers of Pain Therefore, the mechanisms of pain sensitization are complex, and involve changes in nociceptor neurons at both the molecular and cellular level. Furthermore, inflammatory responses can act through growth factor regulation to modulate both the quantity and quality of tissue innervation by nociceptor nerve endings. Inflammation and injury can also lead to changes in the transcriptional profiles of DRG sensory neurons with upregulation of TRP channels or other noxious molecular transducers, allowing previously unresponsive neurons to gain the ability to respond to noxious stimuli. For example, bradykinin is released during inflammation to activate the bradykinin receptor on nociceptors, inducing phospholipase C and protein kinase A signaling, which potentiates TRPA1 opening and pain signaling. Ligand-gated G-protein coupled receptors are often coupled with TRP channel signaling. Na v1.7, Na v1.8, Na v1.9), producing changes in membrane properties, increased action potential firing, and heightened sensitivity to thermal or mechanical stimuli. TRPV1, TRPA1) or modification of voltage-gated sodium channels (e.g. Inflammatory stimuli including cytokines can induce phosphorylation of ligand-gated channels (e.g. Pain sensitization is mediated by multiple mechanisms at both the biophysical and transcriptional levels. Allodynia is a painful response to normally innocuous mechanical or thermal stimuli. Hyperalgesia is increased pain due to a normally noxious stimuli. Pain sensitization can be further categorized as hyperalgesia or allodynia. Pain sensitization is defined as increased responsiveness of nociceptor neurons to their normal or subthreshold afferent imput by the International Association for the Study of Pain (IASP). Therefore, the same cell may be able to respond to multiple sensory stimuli and mediate distinct functional outcomes. Recent work has shown that nociceptors are highly diverse with distinct molecular expression patterns of ion channels, growth factor receptors, G-protein coupled receptors, and neuropeptides. However, these classic groupings are broad and overly simplistic. Aβ and Aδ nociceptor neurons are faster conducting and myelinated neurons, often mediating mechanosensation and mechanical pain sensitivity. C-fiber nociceptor neurons are non-myelinated, slow-conducting neurons that are mostly capsaicin-sensitive and often mediate thermal pain sensitivity. Nociceptor neuron subtypes innervate different epithelial layers in the skin, lung, and gastrointestinal tract, allowing them to interface with environmental stimuli. Specific coupling of nociceptor subtypes with distinct immune cell-types at the molecular level could be a mechanism by which neuro-immune signaling is finely tuned. Nociceptor neurons show remarkable diversity with various cellular subsets mediating heat, cold, and mechanical pain. Nociceptive Neuron Subsets and Pain Sensitization In this review, we discuss recent advances in understanding this bidirectional neuro-immune crosstalk in pain and inflammation. Thus, neural signaling can define the pattern of immune responses and, consequently, contribute to the development of local and systemic inflammatory diseases. Nociceptors in turn release neuropeptides and neurotransmitters that act on innate and adaptive immune cells to modulate their function. Immune cells release mediators that modulate nociceptor neuron activity and pain sensitivity. While pain is critical to induce behavioral changes that lead to avoidance of noxious stimuli, it is also increasingly clear that pain sensation is closely linked to molecular and cellular interactions between the nervous and immune systems. Given the ability of the nervous system to propagate signals within milliseconds, nociceptors are ideally positioned to be first responders to pathogens and tissue injury. Nociceptor nerve terminals express ligand-gated and voltage-gated ion channels including TRPV1, TRPA1, Na v1.7, Na v1.8, and Na v1.9, which are key molecular transducers of these noxious stimuli ( Box 2). Various subsets of nociceptors exist, and can respond to mechanical, chemical or thermal noxious stimuli ( Box 1). Nociceptors are a specialized subset of sensory neurons that mediate pain and densely innervate peripheral tissues including the skin, joints, respiratory, and gastrointestinal tract. Pain is one of four cardinal signs of inflammation defined by Celsus in the 1 st century AD ( De Medicina).
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
